Differential Media:
|
|
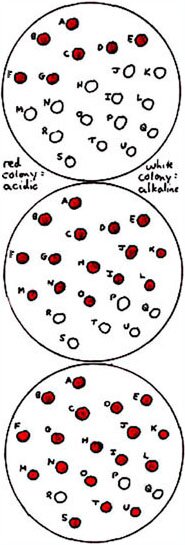
When using a selective plating medium such as MacConkey Agar – which is commonly used for isolating gram-negative rods such as enterics – one also has the ability to differentiate colonies of lactose-fermenting cells from others by the red (acidic) color of the pH indicator. Consider the top plate in the illustration on the right. If one were looking for coliform colonies with MacConkey Agar, one would pick the red colonies (ignoring the white, non-lactose-fermenting colonies) for further testing. Also, if one were looking for an enteric pathogen such as Salmonella or Shigella, one would tend to ignore the red colonies and concentrate on the white ones. Realize that the red vs. white color reactions do not correspond to acid vs. neutral pH reactions. That small amount of ammonium produced from deamination of amino acids (in the peptone) is always going on for organisms that can grow on MacConkey Agar, so an alkaline reaction will be present for a non-lactose fermenter. For lactose-fermenters, the acid produced from fermentation will over-neutralize the ammonium – the net, observed result being the acidic reaction. (For each of the modifications of the medium discussed below, we are maintaining the same colony position for each of the different organisms on the plates shown in the diagram.) If we were looking for a certain enteric organism – let's call it Genus A – that we know does not ferment lactose nor certain other sugars including sucrose, galactose, fructose and mannitol, then we could include these sugars in the medium and look for colonies that show no fermentation whatsoever. The vast majority of enterics would ferment at least galactose and fructose and produce red colonies such as shown in the middle plate on the right. So colonies P Q R S T U would be the ones we would pick for confirmatory testing – to see if any or all were of Genus A. Suppose in studying cultures derived from these six colonies (P through U), we found that three of the strains (P, Q and R) differed in some reactions from those of Genus A and – upon subsequent genetic testing – were determined to belong to an entirely new genus which we will call Genus B. Among the different phenotypic characteristics found for Genus B is the ability to decarboxylate the amino acid lysine. But lysine decarboxylation results in an alkaline reaction, so how can we modify our medium to distinguish Genus B more specifically when lysine decarboxylation would maintain the white, alkaline nature of the colonies?
Our latest modification of the medium results in what we see in the bottom plate. All colonies including those of Genus A (S, T and U) have a red, acidic appearance from fermenting at least one of the sugars in the medium (in the case of Genus A it would be glucose), and the colonies of Genus B show a white appearance due to the net alkaline reaction of lysine decarboxylation overneutralizing the acid produced from the fermentation of the small amount of glucose. Isn't this great fun? The more selective and differential one can make a plating medium, the more likely one may make an interesting discovery – possibly a new species or genus. At UW-Madison, reasoning similar to that shown above went into the formulation of a MacConkey Agar-based medium to help in the detection and isolation of Edwardsiella tarda on which some new biogroups were found and characterized. |
||||||||||||||||||||||||||||
|
These general microbiology pages have copyright by John Lindquist |
Return to the main page of |